Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Ichihara, Akira; Matsuoka, Leo; Kurosaki, Yuzuru; Yokoyama, Keiichi
no journal, ,
Rotational transition probabilities were calculated using the close-coupling method in order to explore the possibility of isotope-selective rotational excitation of LiCl by an optical frequency comb. In the computation, 500 rovibrational states were taken into account in LiCl, and 200 spectral frequencies were included in the frequency comb. The  Li
Li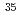 Cl molecule could be rotationally highly excited in gaseous mixture of
Cl molecule could be rotationally highly excited in gaseous mixture of  Li
Li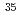 Cl and
Cl and  Li
Li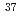 Cl, by tuning spectral frequencies of the comb to rotational transition frequencies of
Cl, by tuning spectral frequencies of the comb to rotational transition frequencies of  Li
Li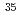 Cl. The isotope selectivity decreases with the increase of the intensity of comb electric field. The vibrational excitation was not induced by the comb employed here.
Cl. The isotope selectivity decreases with the increase of the intensity of comb electric field. The vibrational excitation was not induced by the comb employed here.
Asano, Masaharu; Maekawa, Yasunari; Park, J.*; Takayama, Toshio*; Kudo, Kazuaki*
no journal, ,
no abstracts in English
Saeki, Morihisa; Toshimitsu, Masaaki; Oba, Hironori
no journal, ,
One ton of high-level radioactive waste (HLRW) that is generated from nuclear power plant contains a few kilograms of refractory and platinum-group metals, such as Mo, Zr and Pd. Separation of refractory and platinum-group metals from HLRW contributes to smooth operation of vitrification process. On the other hand, titanium oxide materials adsorb metals by ion-exchange function and micronize metals by photocatalyst function. In this work, we attempted to separate the refractory and platinum-group metals from simulated solution of HLRW by using the titanium oxide nanoparticles, and investigate dependence of recovery efficiency on the particle size.
Sato, Tetsuya; Kaneya, Yusuke*; Sonoda, Nozomi; Asai, Masato; Tsukada, Kazuaki; Toyoshima, Atsushi; Oe, Kazuhiro; Miyashita, Sunao; Nagame, Yuichiro; Sch del, M.; et al.
del, M.; et al.
no journal, ,
In order to determine the first ionization potential (IP) of lawrencium (Lr, Z=103), we have developed a surface ionization type ion-source coupled to a He/CdI gas-jet transport system for the JAEA-ISOL. We measured ionization efficiencies of
gas-jet transport system for the JAEA-ISOL. We measured ionization efficiencies of  Lr and various short-lived lanthanide isotopes by applying the ion-source. We successfully ionized and mass-separated for the first time
Lr and various short-lived lanthanide isotopes by applying the ion-source. We successfully ionized and mass-separated for the first time  Lr ions. From a correlation between the IPs and ionization efficiencies of the short-lived lanthanide isotopes, we estimated the IP of Lr in this work.
Lr ions. From a correlation between the IPs and ionization efficiencies of the short-lived lanthanide isotopes, we estimated the IP of Lr in this work.
Oe, Kazuhiro; Tsukada, Kazuaki; Asai, Masato; Sato, Tetsuya; Toyoshima, Atsushi; Miyashita, Sunao; Nagame, Yuichiro; Sch del, M.; Kaneya, Yusuke*; Lerum, H. V.*; et al.
del, M.; Kaneya, Yusuke*; Lerum, H. V.*; et al.
no journal, ,
We are planning to investigate the redox behavior of element 106, seaborgium (Sg). For the Sg study, we newly developed a continuous dissolution apparatus which dissolves the gas-jet transported nuclides produced in nuclear reactions into aqueous solution. The new apparatus has a hydrophobic membrane filter for separation of aqueous solution from the gas. We investigated the dissolution efficiencies with the apparatus for short-lived nuclides of molybdenum and tungsten which are lighter homologues of Sg. In the meeting, the dependence of the efficiencies on the aqueous- and gas-flow rates will be reported.
Miyashita, Sunao; Oe, Kazuhiro; Toyoshima, Atsushi; Sato, Tetsuya; Asai, Masato; Tsukada, Kazuaki; Nagame, Yuichiro; Sch del, M.; Kaneya, Yusuke*; Omtvedt, J. P.*; et al.
del, M.; Kaneya, Yusuke*; Omtvedt, J. P.*; et al.
no journal, ,
For the experiments of the redox potentials of Sg, rapid separation between different oxidation states of Sg is needed. In this study, solvent extraction of hexavalent Mo and W, as the lighter homologues of Sg, from 1.0 mol/L hydrochloric acid solution into di-(2-ethylhexyl)phosphoric acid (HDEHP), 1-phenyl-3-metyl-4-benzoyl-5-pyrazolone (PMBP), N-benzoyl-N-pheny-hydroxylamine (BPHA) and 4-isopropyl-tropolone (HT) in toluene was carried out. When HDEHP and PMBP were used as extractant, extraction equilibrium of  W was achieved within 5 and 1 hours respectively. On the other hand, when BPHA and HT were used, extraction equilibrium of
W was achieved within 5 and 1 hours respectively. On the other hand, when BPHA and HT were used, extraction equilibrium of  W was quickly achieved within 1 minute in both extractants. Extraction kinetics of
W was quickly achieved within 1 minute in both extractants. Extraction kinetics of 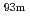 Mo using BPHA and HT was examined. Extraction kinetics of Mo was slower than that of W. Extraction equilibrium of Mo was achieved within 10 by BPHA and 3 minutes by HT. We concluded that HT is a suitable extractant for the Sg reduction experiment.
Mo using BPHA and HT was examined. Extraction kinetics of Mo was slower than that of W. Extraction equilibrium of Mo was achieved within 10 by BPHA and 3 minutes by HT. We concluded that HT is a suitable extractant for the Sg reduction experiment.
Kaneya, Yusuke*; Sato, Tetsuya; Sonoda, Nozomi; Asai, Masato; Tsukada, Kazuaki; Toyoshima, Atsushi; Oe, Kazuhiro; Miyashita, Sunao; Nagame, Yuichiro; Sch del, M.; et al.
del, M.; et al.
no journal, ,
We have developed a surface ionization ion-source as part of the JAEA-ISOL that is coupled to a He/CdI gas-jet transport system to determine the first ionization potential (IP) of heavy actinides. Ionization efficiencies of various short-lived lanthanide isotopes produced in the nuclear reactions were measured with the present system. Obtained results demonstrate that the developed ion-source could be applicable to the measurement of the IP of heavy actinides.
gas-jet transport system to determine the first ionization potential (IP) of heavy actinides. Ionization efficiencies of various short-lived lanthanide isotopes produced in the nuclear reactions were measured with the present system. Obtained results demonstrate that the developed ion-source could be applicable to the measurement of the IP of heavy actinides.
Sekine, Yurina; Fukazawa, Tomoko*; Kajiwara, Yutaro*; Ito, Yu*; Fukazawa, Hiroshi
no journal, ,
no abstracts in English
Kobayashi, Toru; Suzuki, Shinichi; Shiwaku, Hideaki; Okamoto, Yoshihiro; Yaita, Tsuyoshi
no journal, ,
The oxygen-nitrogen hetero-donor ligand PTA has very interesting properties of recognizing the slightly difference of lanthanide ion size, and forming pseudo-enantiomeric complex with boundary at specific element. In this presentation, we will discuss the ion size recognition mechanism based on the detailed structural properties and complexation properties of lanthanide-PTA complex. In addition, novel separation method for specific lanthanide using pseudo-enantiomer formation also will be discussed.
Awual, M. R.; Kobayashi, Toru; Miyazaki, Yuji; Motokawa, Ryuhei; Shiwaku, Hideaki; Suzuki, Shinichi; Okamoto, Yoshihiro; Yaita, Tsuyoshi
no journal, ,